From Initial Discoveries to Market Approval
Each phase is critical, requiring extensive research, significant investment, and regulatory oversight.
From Initial Discovery to Market Approval
Each phase is critical, requiring extensive research, significant investment, and regulatory oversight.
scPharma
Science & Pipeline
We are currently developing a portfolio of products to include novel pharmaceuticals, drug device combinations, and health technologies designed to meet the unique needs of cardiorenal patients, often requiring specialized handling, administration, and monitoring to ensure the highest standards of efficacy and safety.
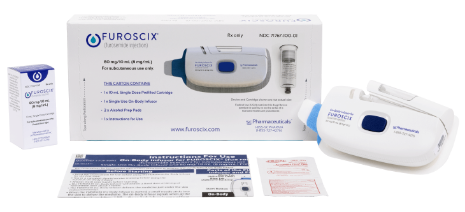
FUROSCIX®
(furosemide injection) 80mg/10mL for Subcutaneous Use.
Preclinical
Phase 1
Phase 2
Phase 3
Phase 4
Supports fluid management in chronic heart failure and chronic kidney disease
INDICATION
FUROSCIX® (furosemide injection), 80 mg/10 mL for subcutaneous use is indicated for the treatment of edema (i.e., congestion, fluid overload, or hypervolemia) in adult patients with chronic heart failure or chronic kidney disease (CKD), including the nephrotic syndrome.
IMPORTANT SAFETY INFORMATION
FUROSCIX is contraindicated in patients with anuria and in patients with a history of hypersensitivity to furosemide, any component of the FUROSCIX formulation, or medical adhesives.
Furosemide may cause fluid, electrolyte, and metabolic abnormalities, particularly in patients receiving higher doses, patients with inadequate oral electrolyte intake, and in elderly patients. Serum electrolytes, CO2, BUN, creatinine, glucose, and uric acid should be monitored frequently during furosemide therapy.
Excessive diuresis may cause dehydration and blood volume reduction with circulatory collapse and possibly vascular thrombosis and embolism, particularly in elderly patients.
Furosemide can cause dehydration and azotemia. If increasing azotemia and oliguria occur during treatment of severe progressive renal disease, discontinue furosemide.
Cases of tinnitus and reversible or irreversible hearing impairment and deafness have been reported with furosemide. Reports usually indicate that furosemide ototoxicity is associated with rapid injection, severe renal impairment, the use of higher than recommended doses, hypoproteinemia or concomitant therapy with aminoglycoside antibiotics, ethacrynic acid, or other ototoxic drugs.
In patients with severe symptoms of urinary retention (because of bladder emptying disorders, prostatic hyperplasia, urethral narrowing), the administration of furosemide can cause acute urinary retention related to increased production and retention of urine. These patients require careful monitoring, especially during the initial stages of treatment.
Contact with water or other fluids and certain patient movements during treatment may cause the On-body Infusor to prematurely terminate infusion. Ensure patients can detect and respond to alarms.
The most common adverse reactions with FUROSCIX administration in clinical trials were site and skin reactions including erythema, bruising, edema, and injection site pain.
Please see the full Prescribing Information and Instructions for Use.
FUROSCIX®
(furosemide injection) 80mg/10mL for Subcutaneous Use.
Preclinical
Phase 1
Phase 2
Phase 3
Phase 4
Under FDA review for potential use in patients with CKD experiencing fluid overload

SCP-111
(Autoinjector)
low-volume, pH-neutral furosemide formulation.
Preclinical
Phase 1
Phase 2
Phase 3
Phase 4
Being developed for potential use in adult patients experiencing edema due to chronic heart failure and/or chronic kidney disease.

We Maximize Commercial Success for Late-Stage Biotech and Pharmaceutical R&D Companies
scPharmaceuticals is the go-to commercialization partner, providing tailored solutions for late-stage cardiovascular and renal R&D companies seeking to bring their products to market swiftly, without the challenges of traditional commercialization approaches.